  By expanding consideration of entropy changes to include the surroundings, we may reach a significant conclusion regarding the relation between this property and spontaneity. When the process is exothermic (Hsystem <0), and the entropy of the system increases (Ssystem >0), the sign of Gsystem is negative at all temperatures. By expanding consideration of entropy changes to include the surroundings, we may reach a significant conclusion regarding the relation between this property and spontaneity. When the process is exothermic (Hsystem <0), and the entropy of the system increases (Ssystem >0), the sign of Gsystem is negative at all temperatures. 
A spontaneous reaction can be fast or slow. Processes that involve an increase in entropy of the system (Δ S > 0) are very often spontaneous however, examples to the contrary are plentiful. Spontaneity- The likelihood of a reaction to proceed without outside intervention. (Show more) thermodynamics, science of the relationship between heat, work, temperature, and energy. So the two conditions for a reaction to be spontaneous are: tendency to achieve minimum energy tendency to achieve maximum randomness. 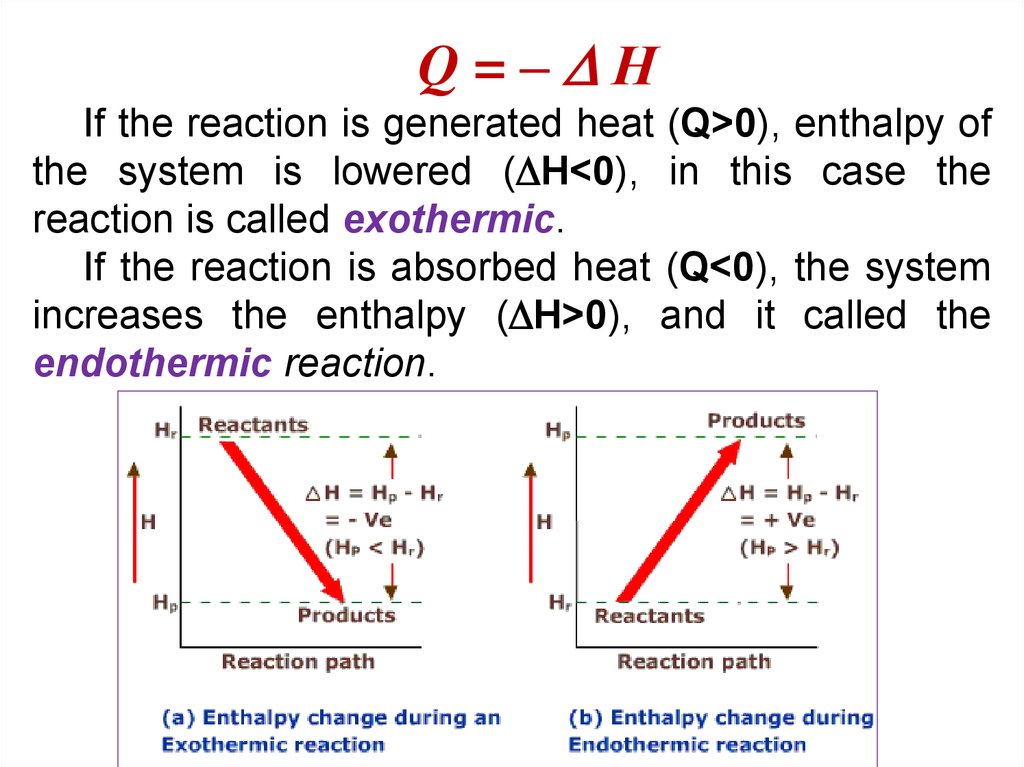
In the quest to identify a property that may reliably predict the spontaneity of a process, we have identified a very promising candidate: entropy. Considering entropy (S) as a thermodynamic parameter, the criterion for the spontaneity of any process is Here the volume of gas increase from V1 to V2 at. Calculate entropy changes for phase transitions and chemical reactions under standard conditions.1st Law Energy can be converted from one form. latexDelta Uq+w0+00/latex The spontaneity of this process is therefore not a consequence of any change in energy that accompanies the process. State and explain the second and third laws of thermodynamics is negative when work is done by the system on the surroundings. The first law of thermodynamics confirms that there has been no change in the system’s internal energy as a result of this process. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
